Ultherapy in New York
- 1. A non-invasive skin lifting
- 2. Ultherapy for face
- 3. Ultherapy for stomach
- 4. Ultherapy recovery and results
- 5. Ultherapy results
- 6. Ultherapy side effects
- 7. Best Ultherapy doctor in NYC
- 8. Ultherapy reviews for Dr. Rokhsar
- 9. Ultherapy FAQ
- 10. Request an Appointment
- 11. New York Office Locations
One of the major hallmarks of time and aging on the skin is the development of looseness and skin laxity. Elements of nature along with gravity and intrinsic aging results in sagging and drooping of the skin over time. With age, our skin doesn’t feel as firm as it once used to. Besides developing lower face loose skin, called jowls, the neck can also become loose commonly referred to as the “turkey neck”. This name is affectionately coined for the sagging of excess skin under the chin and along the neck and jawline. Factors that can speed up the development of turkey neck are genetics, sun exposure, and smoking.
Ulthera, also known as Ultherapy 1, is a non-surgical procedure that tightens the skin of the face and neck using focused ultrasound resulting in a nonsurgical facelift and neck lift with virtually no downtime. Some patients may experience mild swelling post-treatment, while the majority heal with no swelling. The connective tissue begins to immediately tighten, showing visible results in 30 days but taking up to three to six months to fully respond to the treatment.
Dr. Cameron Rokhsar in NYC and LI has been using the patented Ulthera device since 2009 3 to perform non-invasive brow lifts, facelifts, and neck lifts, being the first Ultherapy doctor in the tristate New York area and one of the first in the nation. As a double board-certified dermatologist and dermatologic surgeon and a fellowship-trained laser surgeon, he is one of the most experienced physicians in the world performing the Ultherapy procedure 2. To learn more about the Ultherapy treatment in NYC, contact one of Dr. Rokhsar’s offices in Manhattan or Long Island to book a consultation.
The Ultherapy Treatment involves placing zones of focused ultrasound heat in various deep layers of the skin to lift and tighten loose skin without cutting tissue. Aside from lifting and tightening, Ultherapy also stimulates new collagen production, which helps in maintaining a youthful appearance. Within hours, your body’s healing responses will be signaled to gently and gradually restore skin and underlying tissue and start the tightening journey.
Best of all, Ultherapy requires no preparation or recuperation! Lifting usually takes place primarily over the first two to three months. Patients can immediately return to normal activities, though the skin may appear flushed or patients can experience mild swelling, tingling, or tenderness for one to two days. Ultherapy has been featured in Harper’s Bazaar, Vogue, Marie Claire, The Rachael Ray Show, The Doctors, Elle Magazine, The View, More Magazine, Good Morning America Health, and many other news and media outlets. Dr. Rokhsar treats the face and neck, the lower eyes, and above the brows in the forehead for a non-surgical brow lift. Another common treatment area is the chest. Dr. Rokhsar has also been an early clinical investigator for the Ulthera device, having investigated the machine’s tightening potential in various areas of the face and body through formal clinical trials. The results of his Ulthera studies have been published in major scientific journals.
A non-invasive solution for skin lifting and toning
Dr. Rokhsar was the first physician in the NYC and LI area to offer the Ultherapy® procedure for lifting and toning loose, lax skin 4 – without surgery! The Ulthera device uses a well-known energy source, ultrasound, in a highly focused energy profile, to strengthen and tighten your skin from the inside out, in a completely non-invasive way.
Dr. Rokhsar was among the first physicians in the world to offer the Ultherapy procedure for skin tightening. Dr. Rokhsar has been involved in multiple research projects on the subject of skin tightening with Ultherapy.
After just one treatment, with no downtime, your skin will gradually exhibit a more refreshed, lifted appearance. Ultherapy provides a high degree of control, consistency, and precision not previously seen with other tissue-tightening procedures. While many areas of the face and neck can be treated, another popular area includes the brows. Ulthera is the only device FDA-approved for a non-surgical brow lift. Ultherapy lifts the brow, reducing excess skin on the lids, and opens up the eyes to provide a refreshed and younger appearance.
Unlike other non-surgical procedures, Ultherapy can treat not only the skin itself but also the deep foundation below the skin that is typically addressed in cosmetic surgical procedures like the facelift. This initiates your body’s collagen-rebuilding process without disturbing the surface of your skin. How? The Ultherapy system’s ultrasound energy (DeepSEE) allows Dr. Rokhsar to see the layers of targeted soft tissue 5 on the ultrasound screen, to treat them with unprecedented precision.
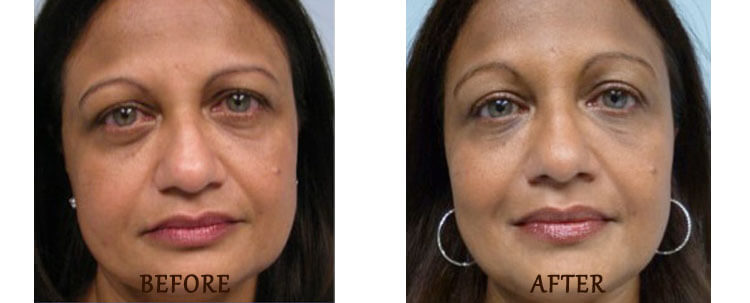
Ultherapy for face
Ultherapy is a non-invasive procedure that uses high-focused, intense ultrasound energy to stimulate collagen growth, and tighten and lift the skin of the face and neck. It is a popular alternative to surgical facelifts, as it requires no downtime with minimal discomfort. During the procedure, ultrasound waves penetrate the skin beneath the surface to reach the deep layers of tissue, where they heat the tissue and stimulate collagen production, resulting in skin tightening and lifting.
While Ultherapy can provide significant skin tightening and lifting benefits, it may not be the right choice for everyone. Individuals with excessive sagging skin may require a more invasive procedure, such as a surgical facelift, or Thermitight radiofrequency skin tightening, to achieve their desired results. Additionally, some patients may experience mild discomfort during the procedure, and the treated area may be sensitive and slightly swollen for a few days post-treatment.
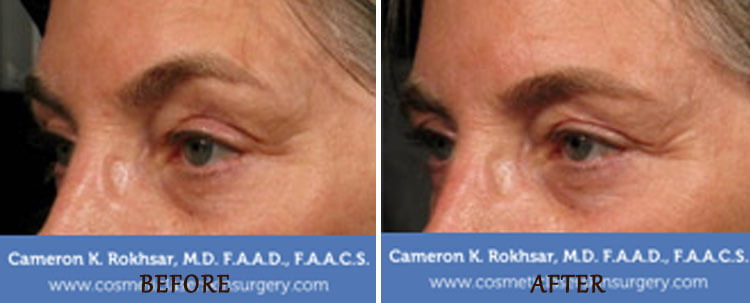
Ultherapy for eyelids
Ultherapy can be used to lift and tighten saggy or droopy skin of the eyebrows but not the upper eyelid without the need for surgery. The lower eyelids and crow’s feet can be treated on the bone. Ulthera can not be used directly on the upper eyelid skin. Dr. Rokhsar prefers to use the CO2 laser for upper eyelid tightening. The results of the CO2 laser for upper eyelid tightening is as good as a surgical eyelift in Dr. Rokhsar’s hands.
Ultherapy works by targeting the deeper layers of skin with ultrasound energy, which stimulates collagen production to tighten the skin. The procedure is non-invasive, which means no incisions are required. Ultherapy generally takes about 2 to 3 hours to perform if done properly.
Ultherapy can provide natural-looking results that improve over time, but the results may not be as dramatic as those achieved through surgery, such as blepharoplasty or surgical brow lifts. However, Ultherapy is a safe and effective option for those who want to improve the appearance of the positions of their eyebrows without undergoing surgery.
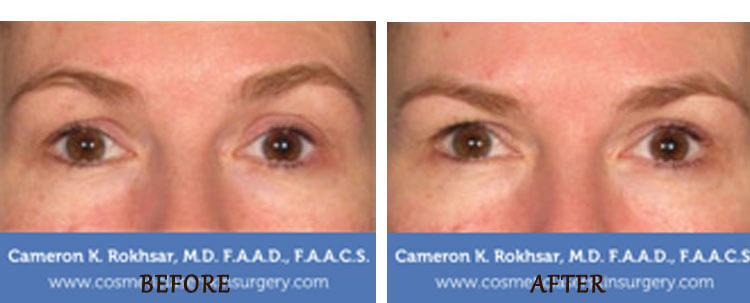
Ultherapy for under eyes
Ulthera is an FDA-approved treatment for lifting and tightening the skin around the eyes, including the under-eye area as long as the treatment is done on the bony orbital rim. This means that the treatment can not be done close to the lower eyelid margin or on the upper eyelids. Unlike other skin tightening procedures that involve cutting or penetrating the skin, Ultherapy bypasses the outer layer of skin and delivers ultrasound energy at about 3 and 4.5 mm under the skin to gently heat the tissue and stimulate collagen production. This helps to improve the appearance of fine lines, wrinkles, and sagging skin around the eyes.
While Ultherapy can be effective for improving the appearance of the under-eye area, it may not be the best option for everyone. In some cases, fillers such as Restylane or resurfacing with the CO2 laser may be more appropriate for addressing under-eye bags, wrinkles, and hollowness.
Ultherapy for brow lift
Ultherapy is an effective alternative to surgical options. The Ulthers device is the only FDA-approved procedure for a non-surgical brow lift. The procedure can tighten and lift the area around the brow and help smoothen wrinkles on the forehead and upper eyelids in a natural way. Results are typically seen gradually over time, with most patients needing just a single treatment session to achieve natural-looking results that improve over time.
Ultherapy for jowls
Ultherapy has been shown to be effective in tightening and lifting sagging skin in the lower face and neck area, including the jowls. Patients generally see gradual improvement over several months as the new collagen forms and tightens the skin. However, the degree of improvement can vary from patient to patient, depending on factors such as age, skin quality, and the severity of the jowls.
Ultherapy is a safe and well-tolerated procedure, with few side effects. Some patients may experience minor swelling or redness immediately after treatment, but these symptoms typically subside within a few hours to a few days. If the jowls are moderate to severe, Dr. Rokhsar may recommend treatment with Kybella injections first to shrink the jowls. Kybella is an FDA-approved drug that can be injected into areas of the face and neck to dissolve fat. Dr. Rokhsar has great success improving the appearance of jowls with a combination of Ulthera and Kybella injections.
Ultherapy for neck
Ulthera is FDA-approved for lifting and tightening the neck, chin, face, and brow areas. Ultherapy for the neck is an excellent option for individuals who are experiencing mild to moderate skin laxity in their neck and jawline areas.
During the procedure, a handheld device is used to deliver ultrasound energy to the deeper layers of the skin in the neck area. This energy causes a controlled injury to the tissue, which stimulates the body’s natural healing response and collagen production. Over time, the newly formed collagen provides a tightening effect, resulting in a more youthful and contoured appearance.
One of the benefits of Ultherapy is that it is a non-surgical and non-invasive procedure. It requires no downtime, and most patients can return to their daily activities immediately following treatment. The procedure typically takes 2-2 hours, depending on the area being treated and the individual treatment plan.
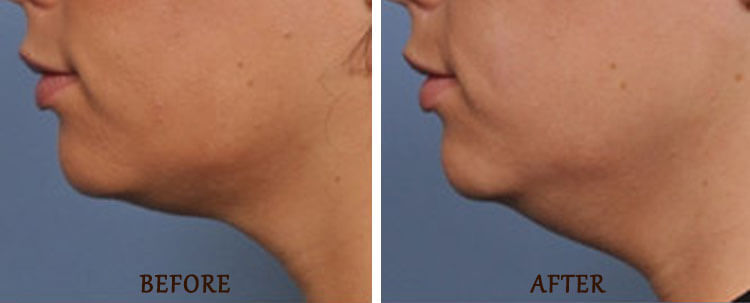
Ultherapy for double chin
The Ultherapy procedure can reduce the appearance of a double chin by tightening the hanging skin the under the chin area. The full benefits of Ultherapy for double chin reduction typically take several months to fully manifest. It is common to experience swelling and redness in the treated area immediately after the procedure, but this typically subsides within a few one to two days. In some cases, patients may experience bruising or tenderness, but these side effects are generally mild and temporary. Most patients have no swelling, or bruising after the treatment.
Most patients only need one Ultherapy treatment to achieve their desired results, and the effects can last for several years. However, the longevity of the results can vary depending on the individual patient’s skin condition and the severity of their double chin. While Ultherapy can be an effective solution for reducing the appearance of a double chin, it is not a substitute for a healthy diet and exercise.
Ultherapy for stomach
While Ultherapy is FDA-cleared for use on the face, neck, and chest, it is also commonly used off-label on other areas of the body, including the stomach, arms, buttocks, thighs, and knees. The treatment has zero downtime, minimal discomfort, and extended collagen stimulation which continues after treatment.
Not everyone is a good candidate for Ultherapy, and a consultation with Dr. Rokhsar is necessary to determine if it is the right option for you. Dr. Rokhsar was involved in developing a protocol for Ulthera treatments off the face.
Ultherapy recovery
As a non-invasive procedure, Ultherapy is a great way to get results without needing to undergo surgery, and the recovery time associated with that. Procedures such as facelifts or neck lifts can require weeks before swelling subsides.
However, Ultherapy recovery is relatively speedy. In most cases; many patients experience no redness or swelling. If there is any swelling, it usually resolves within a day or two. Patients can almost always return to normal activities immediately after their procedure. As a premier Ultherapy provider, Dr. Rokhsar offers Ultherapy treatments at his practices in Long Island and New York.
After just one treatment, you will start feeling the tightening effect of Ulthera on your skin. If you are interested in learning more, please contact New York Cosmetic Skin & Laser Surgery Center today to schedule an appointment!
Ultherapy results
*Results may vary.
Ultherapy side effects
The most common temporary side effects of Ultherapy include redness, swelling, and tingling sensations. These symptoms typically subside within a few hours to a few days after treatment. Mild bruising may occur in some cases, but this usually resolves within a few days to a week. Other less common temporary side effects include nerve injury or palsy and temporary local muscle weakness. These usually resolve within a couple of months. A knowledge of anatomy is very important when performing the Ulthera procedure. That’s why, it is best to have your Ultherpy procedure performed by a board-certified dermatologist to minimize the risk of nerve injury.
Serious complications associated with Ultherapy are rare, but they can occur. These include muscle deterioration, eye herniation, nerve damage, permanent scarring, and vision loss if treated close to the eyes. These risks are extremely rare and can be mitigated by choosing an experienced board-certified dermatologist who is well-trained in performing the procedure.
Dr. Rokhsar is the best Ultherapy doctor in NYC
Dr. Rokhsar was the first Ultherapy doctor in the New York area. He has been performing the procedure since 2009, longer than any other physician. Dr. Rokhsar conducted many of the early clinical trials for Ulthera as a clinical investigator. Dr. Rokhsar is double board certified in dermatology and dermatologic surgery. He is a professor of Dermatology at Mount Sinai Hospital where he teaches dermatology residents in cosmetic dermatology. Dr. Rokhsar is a fellowship-trained laser surgeon, using over 40 lasers and energy-based devices at his laser centers in New York and Long Island. The knowledge of facial anatomy is crucial in performing the Ultherapy procedure to minimize the risk of nerve injury. This is why you should trust your face to a board-certified dermatologist who is experienced in lasers and energy-based devices. Dr. Rokhsar performs all Ultherapy procedures himself for your safety.
To learn more about Dr. Rokhsar’s Ulthera treatment in NYC or LI, contact his offices and book a consultation today.
Ultherapy reviews for Dr. Rokhsar
Ultherapy FAQ
How is the Ultherapy procedure different from other cosmetic procedures?
Ultherapy utilizes ultrasound technology to specifically target and strengthen the deep tissue layers that plastic surgeons address in a facelift or neck lift surgery. Ulthera was the first ultrasound procedure to use this energy in a focused manner to target deep layers of the skin for precise skin tightening. It is completely non-invasive. Unlike radiofrequency microneedling devices like Morpheus8, Ulthera does not pierce your skin with needles. Therefore there is no bleeding or bruising. Swelling is also minimal compared to radiofrequency microneedling procedures like morpheus8. Ultherapy usually requires only one session. In contrast, radiofrequency microneedling like Morpheus 8 requires at least 3 sessions once a month.
How soon will I see the results?
Although people may start noticing a difference in the quality and tightness of their skin within one to two weeks, full results take three months to appear. This is because the process of collagen remodeling can take 3-6 months in response to any laser or energy-based skin tightening procedure.
How long do the results from Ulthera last?
Though the duration of results can vary based on factors like genetics, weight loss, sun exposure, and smoking. Most patients report that the tightening effect from the procedure lasts one to two years.
Is there any downtime after Ulthera treatment?
Because Ulthera is a non-surgical treatment, and the top layer of skin is not injured, there is no medical downtime after the procedure. From start to finish, the procedure only takes about two to three hours, and patients generally return to their normal routines immediately after. Side effects include redness, swelling, tingling, and tenderness. These are usually mild and generally resolve quickly within a day or two. Some patients get no visible swelling. Bruising is also rare. Patients do experience mild tenderness on the jawline area when touching it for one to two weeks.
Who is a good candidate for Ultherapy?
As with many non-invasive cosmetic surgery procedures, Ultherapy is a great option for many patients looking for results without a scalpel. The best candidates for Ultherapy usually possess the following characteristics:
- You have slight to moderate skin laxity.
- This skin feels less firm than you would like it to be.
- Loose skin is located around the chin, neck, lower face, or around the eyes.
- You want a subtle, but noticeable, change in the skin of your face and neck.
- You want the skin around your face and neck to look firmer and more youthful.
- You have realistic expectations about what this procedure can accomplish.
- You understand this procedure is not a substitute for a surgical facelift.
- You are generally healthy enough for this procedure.
- You looking for a non-surgical brow lift option.
Dr. Cameron Rokhsar is an experienced cosmetic surgeon whose New York Ultherapy patients are thrilled with their results because he performs the procedure himself for your safety. Dr. Rokhsar has been doing Ulthera since 2009, longer than any other physician in the New York or Long Island area. He has published research articles on the use of Ultherapy for skin tightening of various parts of the body.
How effective is Ultherapy?
Ultherapy is a non-surgical skin tightening procedure that uses high-intensity focused ultrasound (HIFU) energy to heat the tissue below the surface of the skin, stimulating the body’s natural collagen growth. Ultherapy can improve skin laxity and reduce wrinkles, fine lines, and sagging skin in the brow, neck, jowls, lower face, and chin areas. The results from Ultherapy last for approximately 1-2 years.
Dr. Rokhsar is among the first Ulthera doctor in the United States since it became available in 2008. Dr. Rokhsar was the first physician in the New York area to perform the Ultherapy procedure. New York Cosmetic, Skin & Laser Surgery Center headed by Dr. Cameron Rokhsar in New York and Garden City, was a study site for Ultherpy. Our Centers have published scientific articles in major scientific journals on the Ultherapy procedure.
Ultherapy is FDA-cleared to lift and tighten skin on the neck, chin, lower face, jowls, and brow, as well as improve lines and wrinkles on the décolletage. It is also effective for lifting the eyebrow and tightening skin around the chin and neck. In multiple scientific studies, Ultherapy has been found to be very effective in lifting the skin. Dr. Rokhsar has been performing the Ultherapy procedure since 2008 and has years of experience with it.
How to prepare for Ultherapy?
Here are some tips on how to prepare for Ultherapy:
- Schedule a consultation: The first step in preparing for Ultherapy is to schedule a consultation with Dr. Rokhsar to assess your skin, address your concerns, and determine if you are a good candidate for Ultherapy.
- Stop blood-thinning medications: If you are taking medications that thin your blood, such as aspirin, ibuprofen, or blood thinners, stop taking them a few days before your Ultherapy treatment to reduce the risk of bruising. If you are unsure about which medications to discontinue, speak to Dr. Rokhsar or any member of our medical staff. If you can not stop your blood thinners, it is still ok to do the procedure. You may just have a higher chance of getting bruising.
- Avoid harsh skincare products: Three days before your Ultherapy procedure, avoid using topical products containing isotretinoin (e.g. Retin-A®, Renova, etc.), glycolic acid, or salicylic acid, as they can irritate the skin. Also, remove makeup and other skincare products from the target areas before arriving at your appointment.
- Maintain regular routines: You can maintain your regular eating, drinking, exercise, and work routines before and immediately after the Ultherapy procedure. There is no need for any special preparation for Ultherapy.
How many Ultherapy treatments are needed?
Typically, results from Ultherapy last approximately 1-2 years for patients who are good candidates for the procedure. Patients may wish to repeat the procedure later on. In most cases, one appointment of roughly two hours in length achieves excellent results. The treatment is safe for all skin types and tones including black, brown, and Asian skin, requiring no downtime. Ultimately, the best way to determine if you are a good candidate for Ultherapy is to consult with Dr. Rokhsar who can evaluate individual needs and recommend a personalized treatment plan.
How painful is Ultherapy?
Ultherapy is a non-surgical skin tightening procedure that uses high-intensity ultrasound energy to stimulate the body’s natural collagen growth and improve the appearance of skin sagging, wrinkles, and drooping in various areas like the face, neck, chest, and brow area.
The level of pain experienced during Ultherapy varies from person to person, and it also depends on the treatment area and individual pain tolerance. Dr. Rokhsar administers medications in the office that help manage pain and keep patients relaxed throughout the procedure. We also offer Nitrous Oxide gas, similar to laughing gas, for additional pain control. Some patients may also feel slightly tender for a few weeks, while others may experience temporary redness, swelling, or itchiness immediately after the treatment.
What to avoid after Ultherapy?
After the treatment, there are a few things that you should avoid to maximize the results and reduce any potential side effects.
It is recommended to avoid hot showers, saunas, and direct sunlight for the first 24 hours after Ultherapy. Heat can increase swelling and redness, which are common side effects of the treatment. Unprotected sun exposure should also be avoided for about two weeks after the treatment as the sun’s UV rays can accelerate the skin’s aging process, leading to spots and premature wrinkles. It is advisable to limit sun exposure in general and use sunscreen with an SPF of 30 or greater while going out. Reapply it after every two hours if you have to stay longer in UV rays.
It is also important to avoid wearing makeup for at least 24 hours after the treatment as makeup products can irritate the treated area and cause redness and swelling. Similarly, avoid scratching or vigorously rubbing your skin after the Ultherapy treatment, as the skin is sensitive, and scratching can cause rashes or scars.
Strenuous exercise and hot saunas should be avoided as increased blood flow to the head may increase the risk of swelling and bruising, but it will not affect the results of the treatment. Also, blood-thinning medications and supplements like aspirin and anti-inflammatories should be avoided before the treatment as they can exacerbate bruising. If you are unsure which medications should be avoided, please ask Dr. Rokhsar or any member of our medical staff.
New York Office Locations
Upper East Side Manhattan Office
121 East 60th Street, Suite 8AB New York, NY 10022
(212) 285-1110
Long Island Office
901 Stewart Ave, Suite 240, Garden City, NY 11530
(516) 512-7616
Request an Appointment
References
- Ultherapy: What You Need to Know. Available: https://www.realself.com/nonsurgical/ultherapy.
- FDA approval of Ulthera. Available: https://www.accessdata.fda.gov/cdrh_docs/pdf13/k134032.pdf.
- WHO SAYS ULTHERAPY® BOOSTS YOUR COLLAGEN? OH RIGHT, SCIENCE. Available: https://ultherapy.com/.
- Everything You Need to Know About Premature Aging. Available: https://www.healthline.com/health/beauty-skin-care/premature-aging.
- What Are Soft Tissue Injuries?. Available: https://www.verywellhealth.com/what-are-soft-tissue-injuries-4144746.



 Dr. Rokhsar was chosen by
Dr. Rokhsar was chosen by 






