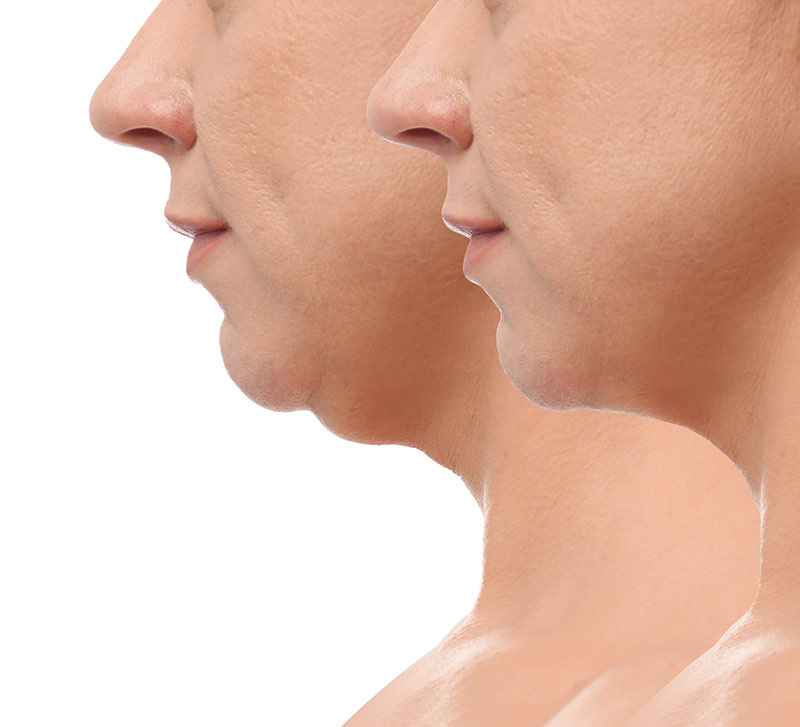
Health Benefits of Protein for Skin, Hair, & Nail Growth
Curiosity About Protein and Hair/Skin/Nail Health: People are interested in protein’s role in hair, skin, and nail health because these body parts are composed of proteins, mainly keratin. Adequate protein intake can support their strength, growth, and repair. Types of Protein: Complete Proteins: Contain all essential amino acids; found in animal products like meat, dairy, […]
Read Article

 Dr. Rokhsar was chosen by
Dr. Rokhsar was chosen by