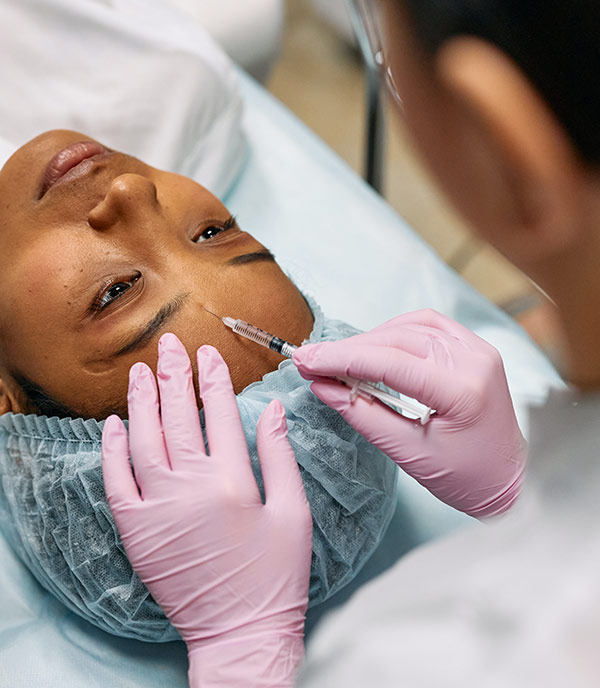
Botox has been used for over a decade as a reliable anti-wrinkle treatment, and now the Food and Drug Administration (FDA) has approved of its use for crow’s feet which are the wrinkles that form next to the eyes. The injected drug utilizes purified Botulinum toxin A to interrupt the line of communications between the brain’s signal to contract a muscle and the muscle tissue itself, ultimately stopping the repetitive movements that cause fine lines and wrinkles in the first place.
Dermatologists and cosmetic surgeons have been using Botox to treat crow’s feet since its release, but now the FDA has officially confirmed its effectiveness as a temporary treatment for crow’s feet. It is the first drug approved to treat wrinkles that form outside the edges of the eyes. It has been proven safe because the botulinun doesn’t leave the injection site, a fear many patients have when they seek out a non-surgical wrinkle treatment.
In two clinical efficacy and safety studies, almost 1,000 participants were treated with moderate to several wrinkles next to the eyes and randomly given either Botox or a placebo. Researchers found that those treated with Botox has a greater improvement and has safe, lasting results.
Botox injections were first approved for the treatment of frown-line wrinkles that form between the eyebrows and have proven to be a successful treatment, especially considering its popularity. Since 2007, Botox has been ranked as the most common cosmetic treatment with $4.6 million procedures performed in the United States, according to the American Society of Plastic Surgeons.
“Botulinum toxin injections are excellent options to treat crow’s feet as well as the prevention of crow’s feet,” says fellowship-trained cosmetic surgeon, Dr. Cameron Rokhsar.
Check in tomorrow to learn more about Botox and the treatment of crow’s feet!

 Dr. Rokhsar was chosen by
Dr. Rokhsar was chosen by 






